 When an organic molecule absorbs infrared light as energy, that causes the chemical bonds to stretch (or bend or rotate depending on how much energy is put in). 
In this case, the reduced mass of the C-H system, 12*1/(12+1) = 0.9, is less than that of the C-C system, 12*12/(12+12) = 6, so the higher mass C-C system has a relatively lower stretching frequency. These two systems have the same reduced mass, each with two carbon atoms, but the stronger C=C double bond has a larger force constant, therefore the stretching frequency of C=C is faster/higher. To extend this to a chemical bond, consider the simplified two-body examples below. From this relationship, we can further conclude that (1) a system with a larger reduced mass will have a lower stretching frequency (the spring will stretch and contract slower if it has heavier masses on each end), and (2) a system with a larger force constant will have a higher stretching frequency (a tighter-wound spring will contract faster after being stretched). Therefore, we can model the stretching or oscillations of this bond with the equation derived from physics, ν = 1/(2π)*√(k/μ), where ν = the frequency of the oscillation (how fast the bond is stretching and contracting), k = the force constant (strength of the bond), and μ = the reduced mass of the system, m 1m 2/(m 1+m 2). 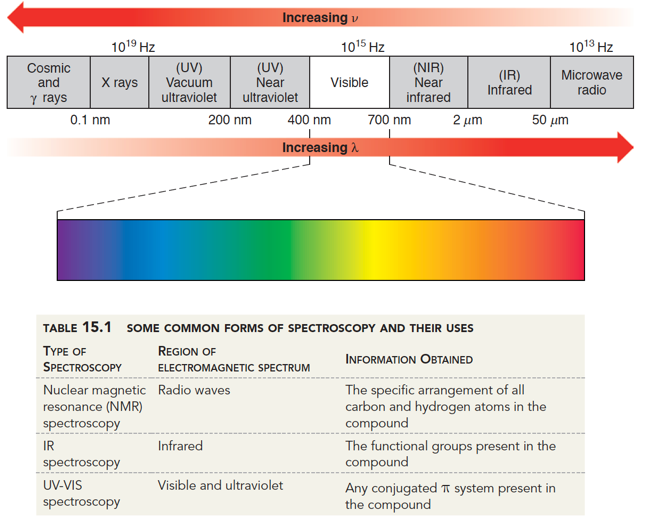
An analogy often used for a chemical bond between two atoms is a spring with a mass at each end. 
By interpreting the frequencies of infrared light absorbed by a molecule, one can identify bond-stretching motions and therefore gain structural information such as what type of functional groups and chemical bonds are present.Ībsorption of infrared radiation by an organic compound causes bonds within that molecule to be stretched, and this phenomenon can be used to gain information about the molecular structure. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
